Evaluation of Local Anti-Stress Agents and Multi-strain Probiotic Bacteria in Transporting Catfish (Clarias anguillaris) Fingerlings
1Department of Animal Health and Production, College of Veterinary Medicine, Joseph Sarwuan Tarka University, Makurdi, Benue State, Nigeria.
2Department of Fisheries and Aquaculture, College of Forestry and Fisheries, Joseph Sarwuan Tarka University, Makurdi, Benue State, Nigeria.
*Correspondence
Edna Ebele OdoEmail:
odoedna@gmail.comAbstract
This study was undertaken to evaluate the anti-stress effect of salt (5g/l), multi-strain probiotic bacteria, and palm oil on the changes in water quality and the survival rate ofClarias anguillaris fingerlings. Six thousand seven hundred and fifty fingerlings were purchased in a homestead pond, acclimatized, and transported. Results showed that the temperature increased progressively during transportation but decreased after the journey, with values ranging from 27.2ºC to 28.3ºC. All the pH values were within the optimum range of 6.5-8.5, except the group treated with salt (T2). There was a steady decline in the dissolved oxygen content of some of the groups during the journey, with T3 recording the highest value of 4.17mg/l and T2 recording the least of 3.07mg/l compared to the control after the journey. There was a progressive increase of total dissolved solids and EC in T1, T3, and T5 during the journey, with T2 recording the significantly (p<0.05) highest value of 7.2 μS/cm and T5 recording the least value of 0.2 μS/cm compared to T1. Ammonia and nitrite concentrations similarly increased steadily across all the groups throughout the journey. The nitrite concentration varied across all the groups during the journey, with T5 recording the highest value of 17.9mg/l compared to T1, which recorded a value of 6.37mg/l. T3 recorded the lowest mortality rate of 2.9% while T1 recorded the highest rate of 42.8%. Based on the results obtained from this study, the multi-strain probiotic bacteria-treated group performed better based on survival and physicochemical parameters, followed by the group treated with salt.
Keywords
Evaluation, Anti-Stress, Multi-strain, Probiotics, Transportation, Fingerlings.
Authors’ Contribution
KOD designed the study, ABN involved in collection of samples and EEO reviewed the work; all authors carried out the field work; wrote and revised the paper.
Citation
Kolndadacha, O.D., Odo, E.E., Ngoyima, A.B., 2025. Evaluation of Local Anti-Stress Agents and Multi-strain Probiotic Bacteria in Transporting Catfish (Clarias anguillaris) Fingerlings. Adv. Fish. Vet. Sci., 1(1): 28-40.
Introduction
Fish represents an important source of protein and micronutrients for many households in Nigeria (Saba et al., 2024). Fish is now the cheapest source of animal protein taken by the ordinary Nigerian, accounting for nearly half of total protein intake (Oluwatobi et al., 2017). Fish farming in urban settings has played an important role in contributing to food supply and the livelihoods of urban inhabitants (Keizire, 2006). Fish is regarded as the best source of animal protein, high in amino acids, and low in cholesterol (Hadyait et al., 2018; Iqbal, 2025; Iqbal and Ashraf, 2025; Shamsan et al., 2025). Furthermore, fish farming is an important way of supplying minerals such as calcium, iron, iodine, and vitamins (Iqbal and Ashraf, 2025; Prein and Ahmed, 2000). Aquaculture production generates revenue through its production, processing, and marketing operations (Gonzalez Parrao et al., 2021; Prein and Ahmed, 2000).
Packing and transportation of fingerlings, juveniles, and broodstock in hatcheries causes stress, which results in mortalities and reduced performance (Omeji et al., 2017; Wang et al., 2024). Transportation stress is reported to be one of the primary contributing factors of fish diseases and mortality in aquaculture (Ren et al., 2022). The survival of fish is affected by many factors during transportation, which include handling, crowding, temperature, and water quality (Hadyait et al., 2020; Iqbal and Ashraf, 2020). Activities involved in the transportation of fish, such as handling, confinement, and exposure to sub-optimal water quality, have the potential to create physiological changes in fish due to stress (Shamsan and Al-Jobory, 2018). Many diseases, particularly parasitic infections, pose a danger to fish farming, which is a major source of food and employment in developing nations (Ali et al., 2016; Iqbal et al., 2019; Odo et al., 2023). The higher occurrence of parasites and mycotoxins in fish denotes the potential public health threat (Al-Jobory, 2025; Hefnawy et al., 2019; Yaqub et al., 2017). Because of the affiliation between stress and fish health, many works were conducted to test the effect of additives such as palm oil, non-iodized salt (Idowu et al., 2016), and probiotic bacteria on the survival of fingerlings prior to, during, and after transportation (Raj, 2008).
Palm oil has been used as food and medicine for many years, but the food sector has recently adopted its use (Alhaji et al., 2024; Mba et al., 2015). Although it has little effect on blood cholesterol levels compared to olive and peanut oil, which do not contain cholesterol, its antioxidant content may help to prevent many diseases (Absalome et al., 2020). Previous studies have shown that palm oil has a good effect on growth performance and increases innate immunity in juveniles of Nile tilapia Oreochromis niloticus (Larbi Ayisi et al., 2018). Interestingly, oil can also be used as a sedative in transporting water to aid in alleviating stress and trauma to fish (Falaye et al., 2012). This study was designed to assess and compare the performances of palm oil at two separate concentrations, salt and multi-strain probiotic bacteria, in alleviating transportation stress in catfish fingerlings transported by road.
Materials and Methods
Experimental fish and management
Catfish (Clarias anguillaris) fingerlings were used in this investigation conducted at Joseph Sarwuan Tarka University, Makurdi. The aquaculture attributes of catfish include: ability to withstand handling stress, disease resistance, high growth rate, yield potential, fecundity, and palatability. Six thousand seven hundred and fifty fingerlings with average weight and length of 2.8±0.2g and 2.1±0.1cm, respectively, were purchased from a homestead pond within Makurdi metropolis and harvested into a 100-liter drum containing 40 liters of water before transportation exercise.
Experiment design
A complete randomized design (CRD) was adopted in the study. Three potential anti-stress agents (salt, multi-strain probiotic bacteria, and palm oil) were administered in water used for transporting the fingerlings to assess their influence on the physicochemical parameters of the water and the responses of the fingerlings to the transportation stress. The six thousand seven hundred and fifty fingerlings were distributed randomly into 5 treatments in replicates as follows:
Treatment 1: To the control, only sterile physiological buffer saline (PBS) was added to the transport water.
Treatment 2: To this group, salt was added to the transport water at a dose rate of 5g/l.
Treatment 3: To this group, multi-strain probiotic bacteria (Bacillus subtilis, B. amyloliquefaciens, B. velenzensis) at equal proportions were added to the transport water at a dose rate of 10ml/L of 1010 CFU/ml.
Treatment 4: To this group, 1.5ml/L palm oil was added to the transport water as a lower dose. Treatment 5: To this group, 2.5ml/L palm oil was added to the transport water as a higher dose.
Loading and transportation of fish
The fingerlings were fasted for 24 hours prior to transportation to reduce the chances of vomiting during the journey due to stress that will affect the water quality (Navarro et al., 2016; Sampaio and Freire, 2016). Six thousand seven hundred and fifty fingerlings were distributed randomly into fifteen Jericans of 10 litres, representing 5 treatments in replicates at the stocking density of 75 fingerlings/l. There was no provision of artificial aeration. The fingerlings were placed into the car and moved for a distance of approximately 500km at a speed of 60-80km/hr. The physicochemical parameters of the water were measured before, during, and after transportation. At the end of the journey, the fingerlings were offloaded from the vehicle, and physicochemical parameters were measured. Thereafter, they were left unpacked for 15 hours without changing their water due to the late arrival. At the 15th hour, the last physicochemical parameters were measured, mortalities were counted from each treatment and replicate.
Preparation of Multi-strain probiotic bacteria
Three stored Bacillus strains isolated, identified, and screened for probiotic properties using standard procedures from the skin and intestinal tract of Clarias anguillaris were purchased from a homestead pond in Makurdi metropolitan. The three species successfully selected were Bacillus subtilis, Bacillus amyloliquefaciens, and Bacillus velezensis based on their performance in pH and bile tolerance, digestive enzymes production, mucosal attachment, and gastrointestinal transit survival tests. Stock culture of the Bacillus species was removed from the refrigerator. Each isolate was subcultured on nutrient agar and incubated for 24 hours at 37ºC. Discrete colonies were harvested using a sterilized blade and were homogenized in sterile distilled water. The turbidity was adjusted to correspond to 7 McFarland turbidity standard, which was equivalent to 1×1010 CFU/ml (Ramlucken et al., 2021). All the isolate suspensions were mixed proportionally to obtain an experimental probiotic mixture of three strains.
Physicochemical parameters of the water used in transportation
Seven physicochemical parameters were measured during this investigation, including dissolved oxygen (DO), pH, total dissolved solids (TDS), electrical conductivity (EC), temperature (T oC), ammonia (NH3), and nitrite (NH2) concentrations. The temperature and oxygen concentrations were measured using the Traceable Dissolved oxygen Meter; the pH, TDS, and electrical conductivity (EC) were measured using the HANNA GROCHEK meter; ammonia and nitrite levels were measured using the Lovibond water testing tinto-meter.
Data analysis
All the data obtained were subjected to analysis of variance (ANOVA), and where significant differences exist, the means were subjected to analysis using Duncan’s multiple range tests. Values of p < 0.05 were considered significant.
Results
The results of the seven physicochemical parameters analyzed, which include pH, temperature, dissolved oxygen, total dissolved solids, electrical conductivity, ammonia, and nitrite, are presented below.
Temperature, pH, and Dissolved oxygen (DO) concentration
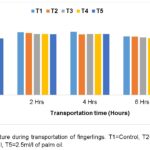
The trend of changes in physicochemical parameters of the water used in transporting catfish fingerlings is presented in Figure 1. There was a progressive increase in temperature across all the groups during the journey; however, there was a fall in temperature after the journey, which was recorded by evening and the following morning. At 0hr, 4hr, and 15hrs, the control group (T1) recorded higher temperatures compared to other treatments.
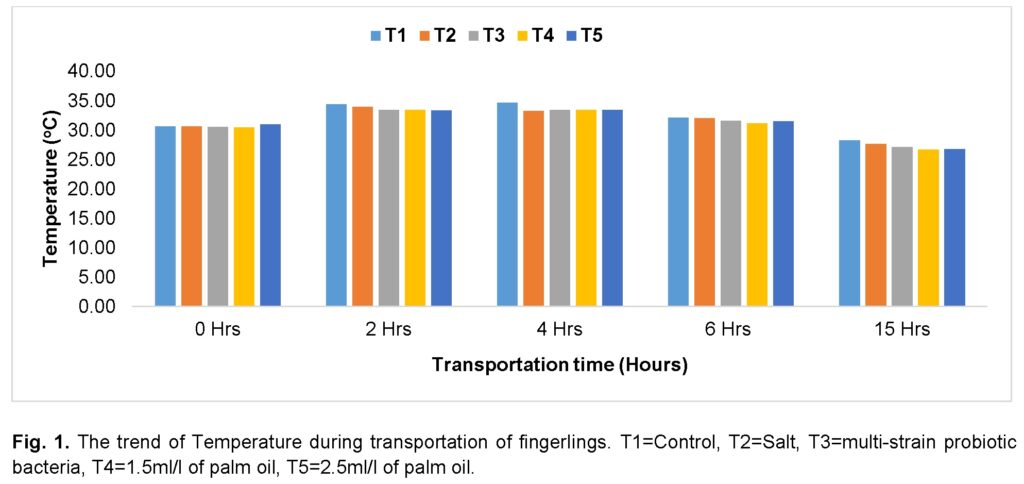
A similar trend of progressive decrease and increase in pH at the beginning and end of the journey, respectively, was recorded as in the case of temperature (Figure 2). The control (T1) had a slightly higher pH at the beginning of the journey, while the group treated with salt (T2) and multi-strain probiotic bacteria (T3) had the lowest pH value. The group treated with salt (T2) recorded a decrease at the 2nd hour, peaked at the 4th hour, and subsequently decreased at the end of the journey. The group treated with multi-strain probiotic bacteria (T3) increased during the journey and recorded the least at the 4th hour, and increased throughout the remaining hours of the journey. The groups treated with palm oil (T4 and T5) recorded similar values at the beginning of the journey, and both decreased at the 2nd and 4th hour. The level further increased at the 6th and 15th hours. The group treated with multi-strain probiotic bacteria recorded the highest value of pH at the end of the journey, followed by the group treated with 1.5ml of palm oil (T4), while the group treated with salt (T2) recorded the lowest value.
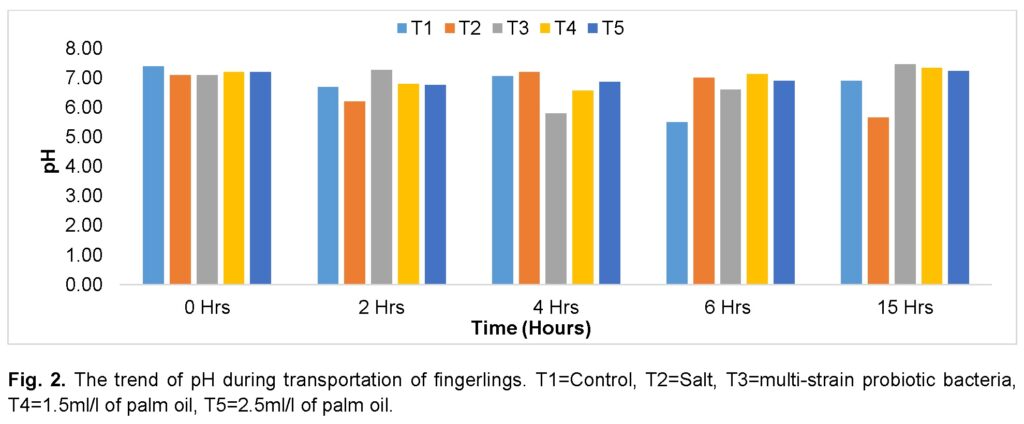
The DO concentration varied across the treatments, with a steady decrease as the journey progressed in the group treated with 1.5ml/l of palm oil (T4). There was a progressive decrease in the groups treated with multi-strain probiotic bacteria (T3) as the journey progressed until the 6th hour, where the value increased slightly to the 15th hour. The groups treated with salt (T2) and 2.5 ml/l (T5) of palm oil recorded the highest and lowest DO, respectively, before the journey. The group treated with 2.5ml of palm oil (T5) recorded the highest value at 2 hours, while the control group (T1) recorded the lowest value. In the control group (T1), there was a progressive decline as the journey progressed and an increase at the 4th hour. The group treated with multi-strain probiotic bacteria (T3) recorded the highest value, while the group treated with salt (T2) recorded the lowest after the journey. At the 15th hour, the group treated with multi-strain probiotic bacteria (T3) had the highest DO, and the group treated with salt (T2) followed (Figure 3).
Total dissolved solids (TDS) and Electrical conductivity (EC)
Figure 4 presents the trend of TDS of the transport water during the journey. There was a progressive increase of total dissolved solids in the control group (T1), the group treated with high dose (2.5ml/l) of palm oil (T5), and the group treated with multi-strain probiotic bacteria (T3) as the journey progressed. The group treated with low dose (1.5ml/l) of palm oil (T4) increased steadily and decreased slightly at the 6th hour and peaked at the 15th hour. The group treated with a high dose (2.5ml) of palm oil (T5) increased slightly during the journey and decreased at the 4th hour till the end of the journey. The group treated with salt (T2) was observed to have significantly (P ≤ 0.05) the highest value of TDS compared to any of the treatments throughout the journey, while the group treated with high dose (2.5ml/l) of palm oil (T5) had the lowest value at the end of the journey.
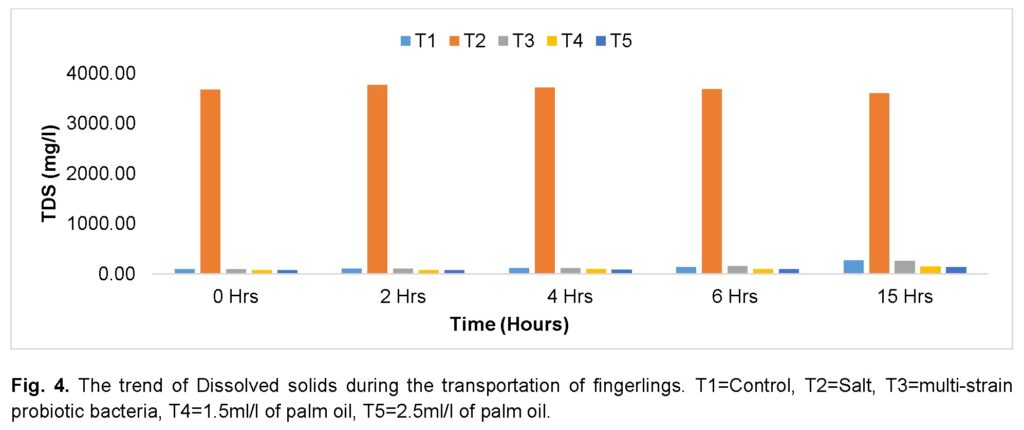
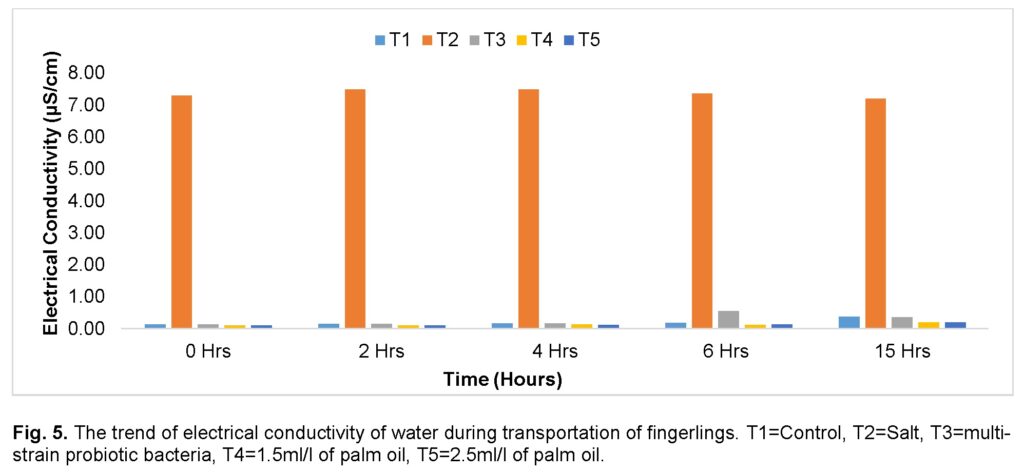
Similarly, significantly (P ≤ 0.05), the highest values of EC were recorded for the group treated with salt (T2) compared to any other treatment throughout the journey (Figure 5). There was a steady increase recorded for the control (T1) and the group treated with high dose (2.5ml/l) of palm oil (T5) throughout the journey. The group treated with multi-strain probiotic bacteria (T3) increased and peaked at the 6th hour and declined at the 15th hour. The group treated with low dose (1.5ml/l) of palm oil (T4) increased moderately during the journey and decreased slightly at the 6th hour, then peaked at the 15th hour. The group treated with multi-strain probiotic bacteria (T3) recorded the lowest values of electrical conductivity before the journey, while the groups treated with high and low doses of palm oil (T4 and T5) recorded similarly low values at the 15th hour.
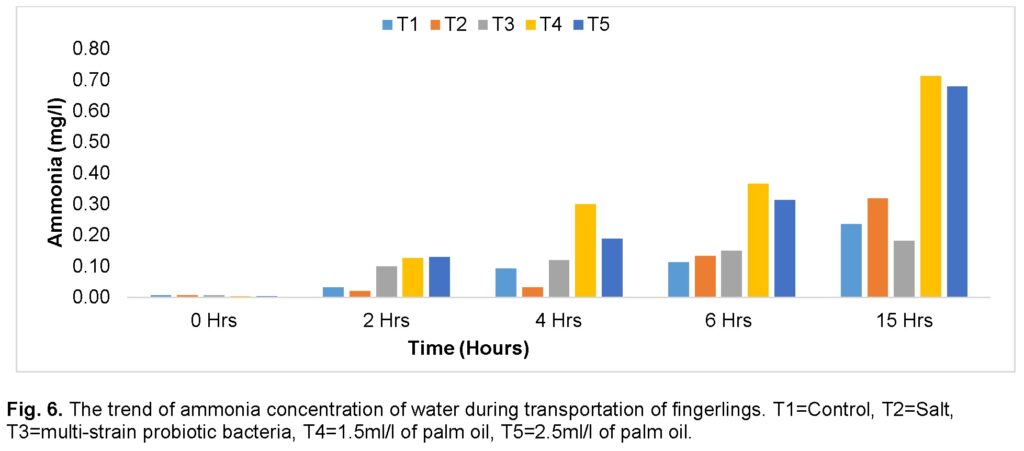
Ammonia and Nitrite
Figure 6 shows the trend of ammonia and nitrite concentration of the transport water during the journey. The ammonia concentration showed a steady increase across all the groups and throughout the journey. Similar values were recorded for all the groups before the journey. The group treated with low dose (1.5ml) of palm oil (T4) recorded the highest value of ammonia concentration followed by the group treated with 2.5ml/l (T5) of palm oil at the end of the journey while the group treated with salt (T2) and group treated with multi-strain probiotic bacteria (T3) recorded similar levels compared with control (T1). At the 15th hour after, the group treated with palm oil 1.5 and 2.5ml, respectively, recorded significantly high levels compared to the control. The group treated with multi-strain probiotic bacteria (T3) had the lowest values.
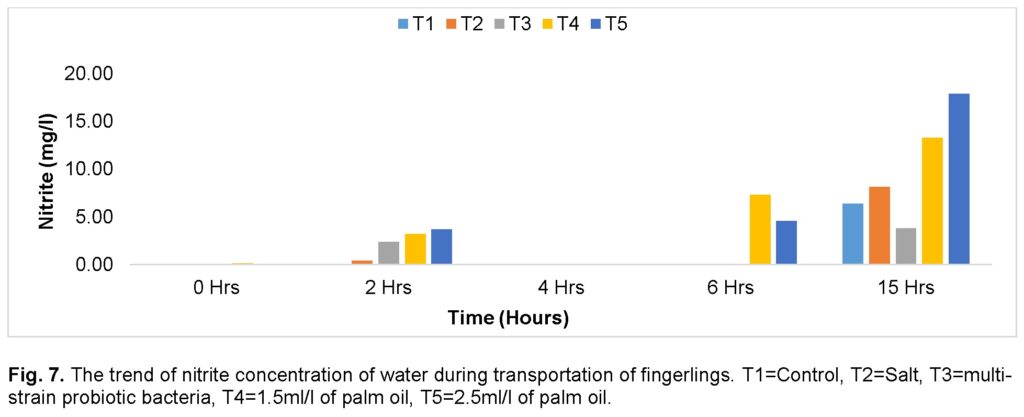
However, the nitrite concentration of the transport water varied across all the groups during the journey (Figure 7). The group treated with high dose (2.5ml/l) of palm oil (T5) recorded the highest value, followed by the group treated with low dose (1.5ml/l) of palm oil (T4) at the end of the journey, while the group treated with multi-strain probiotic bacteria (T3) recorded the lowest value compared to the control group (T1). Similarly, at 15 hours, the group treated with a high dose (2.5ml/l) of palm oil (T5) recorded significantly (P ≤ 0.05) the highest level of nitrite, followed by the group treated with a low dose (1.5ml/l) of palm oil (T4), while T3 (multi-strain probiotic bacteria) recorded the lowest values.
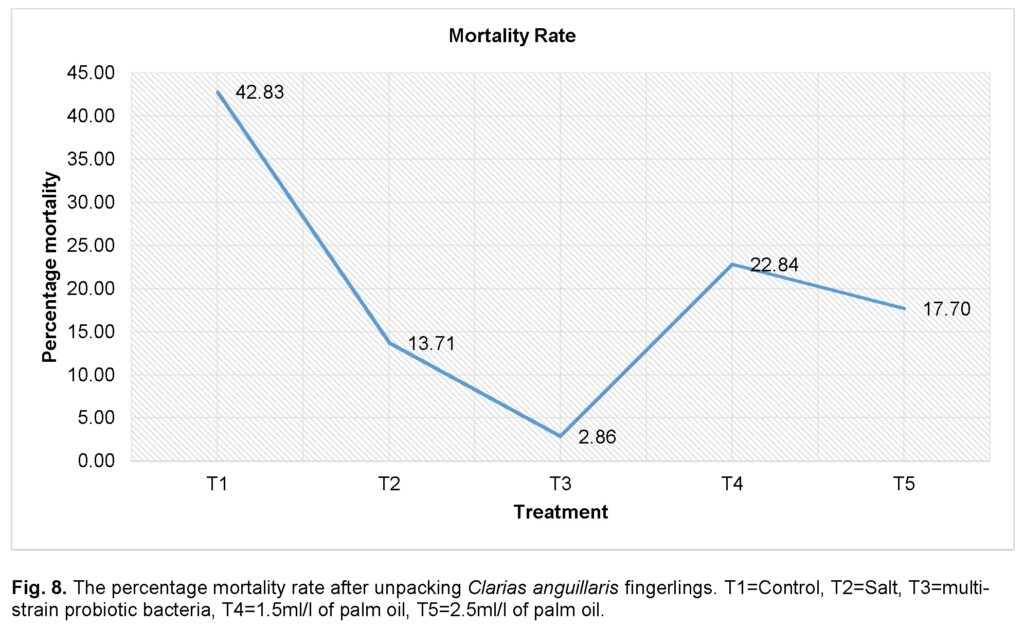
Survivability rate after unpacking Clarias anguillaris fingerlings
Figure 8 shows the mortality rate of the transported fingerling at the end of the transportation exercise. Mortalities were observed across all the treatments at the end of the experiment. The control group (T1) was found to have significantly (P ≤ 0.05) the highest mortality (42.83%), followed by the group treated with low dose (1.5ml/l) of palm oil (T4) (22.88%). The group treated with multi-strain probiotic bacteria (T3) had the least mortality rate (2.86%) compared to all treatments.
Discussion
Transportation stress is reported to be one of the primary predisposing factors of fish disease and mortality in aquaculture (Fang et al., 2023; Rollo et al., 2006). Activities involved in the transportation of fish, such as handling, confinement, and exposure to sub-optimal water quality, have the potential to create physiological changes in fish due to stress. From the results obtained in this study, there was a temperature variation and dissolved oxygen concentration across the groups, with the group treated with probiotic bacteria (T3) recording a steady temperature during the journey with a corresponding level of oxygen concentration. However, after the journey and the following morning, there was a decrease in temperature with a corresponding increase in dissolved oxygen concentration. This agrees with previous studies that reported dissolved oxygen content of transport water decreases with an increase in transport duration, which is attributed to the differing rates of oxygen depletion; consequently, the difference in physiological states of the fish transported in various transport media (Crosby et al., 2005; Wang et al., 2024). Moreover, a change in water temperature has been reported to influence fish activities, especially respiration (Boyd, 1990), which could result in an increase in oxygen consumption (Lindmark et al., 2022; Volkoff and Rønnestad, 2020).
In this study, the group treated with multi-strain probiotic bacteria (T3) had the highest level of dissolved oxygen content after the journey, which agrees with previous studies that reported that probiotics improve water quality, especially Bacillus species (Hlordzi et al., 2020; Mohammed et al., 2025; Tabassum et al., 2021). The rationale is that gram-positive bacteria, especially Bacillus spp., are generally more efficient in converting organic matter back to CO2 than are gram-negative bacteria, which would convert a greater percentage of organic carbon into bacterial biomass or slime. And the group treated with salt (T3) had the second highest dissolved oxygen concentration which agrees with the work of previous investigations which reported that salt reduces physiological stress by decreasing the osmotic gradient between the fish and the transport water thus decreasing the amount of energy the fish would use for osmoregulation (Abou Anni et al., 2016; Crosby et al., 2005; Hana et al., 2023).
In the study conducted, the values of the pH range were between 5.5 and 7.5, with the peak value recorded at the 15th hour in the group treated with multi-strain probiotic bacteria (T3). It has been reported that water pH affects the metabolism and physiological processes of fish and also exerts a considerable influence on the toxicity of ammonia (Edwards et al., 2024; Keremah et al., 2014). The pH is an essential parameter for maintaining life in the aquatic environment, and its stability is very important in fish farming (Mariu et al., 2023; Sanou et al., 2022). Usually, a pH between 6.5 and 8.5 is ideal for biological productivity (Chen et al., 2022). Death is almost certain at a pH below 4 or above 11 (Kane et al., 2015).
There was progressive increase of total dissolved solids in the control group (T1), the group treated with high dose (2.5ml/l) of palm oil (T5) and the group treated with multi-strain probiotic bacteria (T3) as the journey progressed with a corresponding increase in the electrical conductivity of the control (T1) and group treated with high dose (2.5ml/l) of palm oil (T5). The group treated with salt (T2) recorded significantly higher values of TDS than all the other groups throughout the journey. This agrees with previous findings that reported the increase in electrical conductivity would be induced by an increase in TDS levels (Goher et al., 2017). Moreover, the strong positive correlation observed between conductivity and TDS could point out the fact that these two parameters describe the presence of inorganic salts dissolved in solution (Yaka et al., 2020).
Ammonia is produced as a by-product from fish metabolism and is primarily excreted through the gills by diffusion (Shrivastava et al., 2017). In this study, the ammonia concentration showed a steady increase across all the groups and throughout the journey. This agrees with previous reports that an increase in temperature leads to an increase in biochemical reaction rate in fish and consequently increases ammonia excretion by the fish (Volkoff and Rønnestad, 2020). Fish are very sensitive to un-ionized ammonia and need an optimum range of 0.02-0.05mg/l (Keremah et al., 2014; Verma, 2006). Some of the values recorded in this study were above the optimum range during and after the journey. The group treated with low dose (1.5ml) of palm oil (T4) recorded the highest value, while the group treated with multi-strain probiotic bacteria (T3) recorded the lowest value at the 15th hour. Higher levels of ammonia caused reduced growth and gill damage in Channel Catfish (Mitchell and Jr, 2011). However, some of the observed values in this study were higher than the desired range, and this may affect the survival of the fingerlings, which could have caused the high mortality in the control group compared to other treatments with anti-stress agents.
Nitrite is a crucial hazardous chemical for organisms in the aquatic environment that is produced in the process of bacterial nitrification of ammonia or denitrification of nitrate (Gao et al., 2020). Exposure to excessive amounts of nitrite in aquatic settings causes nitrite to accumulate in the bodies of aquatic animals, which can have detrimental toxic consequences (Kim et al., 2018). In this study, the nitrite concentration increased progressively in all groups throughout the journey, with the highest concentration in the group treated with a high dose of palm oil (T5), while the group treated with multi-strain probiotic bacteria recorded the least value at the 15th hour. Nitrite exposure impacts the osmo-regulatory activities, ionic homeostasis, and metabolism of several fish species (Deane and Woo, 2007).
Conclusion
This study reveals that the application of multi-strain probiotic bacteria (Bacillus subtilis, B. amyloliquefacien, B. velenzensis) at 10ml/l of water in transporting fingerlings was good in alleviating the effect of transportation stress with evidence of high survival rate and controlled physio-chemical parameters to suit the fish while the application of salt at 5g/l to transport water also showed a positive effect by recording the second lowest mortality after multi-strain probiotic bacteria. According to the findings of this study, the multi-strain probiotic bacteria-treated group outperformed the salt-treated group in terms of survival and physicochemical parameters.
Acknowledgment
The authors sincerely thank the Department of Fisheries and Aquaculture, College of Forestry and Fisheries, Joseph Sarwuan Tarka University, Makurdi, Benue State, Nigeria, for providing the necessary facilities to complete this research.
Conflict of interest
The authors declare no conflict of interest.
References
Abou Anni, I.S., Bianchini, A., Barcarolli, I.F., Varela, A.S., Robaldo, R.B., Tesser, M.B., Sampaio, L.A., 2016. Salinity influence on growth, osmoregulation and energy turnover in juvenile pompano Trachinotus marginatus Cuvier 1832. Aquac., 455: 63-72.
Absalome, M.A., Massara, C.-C., Alexandre, A.A., Gervais, K., Chantal, G.G.-A., Ferdinand, D., Rhedoor, A.J., Coulibaly, I., George, T.G., Brigitte, T., 2020. Biochemical properties, nutritional values, health benefits and sustainability of palm oil. Biochimie., 178: 81-95.
Al-Jobory, H.J., 2025. A Feast or a Threat? Multi-Mycotoxin Contamination in Sun-Dried Fish, Collected from Al Mukha Markets, Taiz, Yemen. PSM Microbiol., 10(1): 30-43.
Alhaji, A.M., Almeida, E.S., Carneiro, C.R., da Silva, C.A.S., Monteiro, S., Coimbra, J.S.d.R., 2024. Palm Oil (Elaeis guineensis): A Journey through Sustainability, Processing, and Utilization. Foods (Basel, Switzerland), 13(17): 2814.
Ali, S., Akhter, S., Muhammad, A., Khan, I., Khan, W.A., Iqbal, M.N., Umar, S., Ahmed, H., Ali, Q., 2016. Identification, characterization and antibiotic sensitivity of Aeromonas hydrophila, a causative agent of epizootic ulcerative syndrome in wild and farmed fish from potohar, Pakistan. Pak. J. Zool., 48(3): 899-901.
Boyd, C.E., 1990. Water quality in ponds for aquaculture.
Chen, C.-H., Wu, Y.-C., Zhang, J.-X., Chen, Y.-H., 2022. IoT-based fish farm water quality monitoring system. Sensors., 22(17): 6700.
Crosby, T.C., Hill, J.E., Martinez, C.V., Watson, C.A., Pouder, D.B., Yanong, R.P., 2005. On-farm transport of ornamental fish. University of Florida Institute of Food and Agricultural Sciences Extension Publication FA119.
Deane, E.E., Woo, N.Y., 2007. Impact of nitrite exposure on endocrine, osmoregulatory and cytoprotective functions in the marine teleost Sparus sarba. Aquat. Toxicol., 82(2): 85-93.
Edwards, T.M., Puglis, H.J., Kent, D.B., Durán, J.L., Bradshaw, L.M., Farag, A.M., 2024. Ammonia and aquatic ecosystems – A review of global sources, biogeochemical cycling, and effects on fish. Sci. Total Environ., 907: 167911.
Falaye, E., Omoike, A., Folorunso, L., Bello, O., 2012. The effects of palm oil on the physical appearance of Clarias gariepinus during transportation. Int. J. Plant Anim. Environ. Sci., 2(4): 82-89.
Fang, D., Mei, J., Xie, J., Qiu, W., 2023. The Effects of Transport Stress (Temperature and Vibration) on Blood Biochemical Parameters, Oxidative Stress, and Gill Histomorphology of Pearl Gentian Groupers. Fishes., 8(4): 218.
Gao, X.-Q., Fei, F., Huo, H.H., Huang, B., Meng, X.S., Zhang, T., Liu, B.-L., 2020. Impact of nitrite exposure on plasma biochemical parameters and immune-related responses in Takifugu rubripes. Aquat. Toxicol., 218: 105362.
Goher, M., Abdoa, M., Bayoumy, W., El-Ashkar, T., 2017. Some heavy metal contents in surface water and sediment as a pollution index of El-Manzala Lake, Egypt. J. Basic Environ. Sci., 4(2): 210-225.
Gonzalez Parrao, C., Moratti, M., Shisler, S., Snilstveit, B., Eyers, J., 2021. PROTOCOL: Aquaculture for improving productivity, income, nutrition and women’s empowerment in low- and middle-income countries: A systematic review and meta-analysis. Campbell Syst. Rev., 17(3): e1188.
Hadyait, M.A., Ali, A., Bhatti, E.M., 2020. Water Quality Profiles of Fish Farms from the District Attock, Pakistan. PSM Biol. Res., 5(2): 85-91.
Hadyait, M.A., Ali, A., Bhatti, E.M., Qayyum, A., Ullah, M.Z., 2018. Study of proximate composition of some wild and farmed Labeo rohita and Cirrhinus mrigala fishes. PSM Biol. Res., 3(1): 34-38.
Hana, H.N., Abd El-Galil, M.A., Mousa, M.A., Abd El-Lateif, R.S., Osman, A.E., Emam, A.M., 2023. Stress dampening effect of common salt on Oreochromis niloticus during transport. New Valley Vet. J., 3(1): 32-38.
Hefnawy, Y.A., Ahmed, H.A., Dyab, A.K., Abdel-Aziz, A.R., Boules, M.S., 2019. Fish as a potential source of parasites of public health importance in El-Minia governorate, Egypt. PSM Microbiol., 4(2): 44-52.
Hlordzi, V., Kuebutornye, F.K.A., Afriyie, G., Abarike, E.D., Lu, Y., Chi, S., Anokyewaa, M.A., 2020. The use of Bacillus species in maintenance of water quality in aquaculture: A review. Aquac. Rep., 18: 100503.
Idowu, T., Williams, S., Adedeji, H., Sogbesan, O., 2016. Effects of different additives on the survival and haematology of Clarias gariepinus (Burchell, 1822) fingerlings during transportation. Nig. J. Trop. Agric., 16: 65-71.
Iqbal, M.N., 2025. Fish Consumption: Improving Food Security, Nutrition and Livelihoods. PSM Biol. Res., 10(1): 56-58.
Iqbal, M.N., Ashraf, A., 2020. Water Quality a Priority for Profitable Fish Farming. PSM Biol. Res., 5(2): 92-94.
Iqbal, M.N., Ashraf, A., 2025. The Nutrient Content as Indicator of the Validity of Fish as a Healthy Food Source. PSM Biol. Res., 10(1): 53-55.
Iqbal, M.N., Ashraf, A., Iqbal, I., 2019. Parasites of fish: a potential public health concern. PSM Microbiol., 4(2): 53-55.
Kane, S., Qarri, F., Lazo, P., Bekteshi, L., 2015. The effect of physico-chemical parameters and nutrients on fish growth in Narta Lagoon, Albania.
Keizire, B., 2006. Sustainability Impact Assessment of Proposed WTO Negotiations: The Fisheries Sector—Country Case Study: Uganda. Draft for Appendix 9.
Keremah, R., Davies, O., Abezi, I., 2014. Physico-chemical analysis of fish pond water in freshwater areas of Bayelsa State, Nigeria. Greener J. Biol. Sci., 4(2): 033-038.
Kim, J.-H., Kim, J.-Y., Lim, L.-J., Kim, S.K., Choi, H.S., Hur, Y.B., 2018. Effects of waterborne nitrite on hematological parameters and stress indicators in olive flounders, Paralichthys olivaceus, raised in bio-floc and seawater. Chemosphere., 209: 28-34.
Larbi Ayisi, C., Zhao, J., Wu, J.-W., 2018. Replacement of fish oil with palm oil: Effects on growth performance, innate immune response, antioxidant capacity and disease resistance in Nile tilapia (Oreochromis niloticus). PloS one., 13(4): e0196100.
Lindmark, M., Audzijonyte, A., Blanchard, J.L., Gårdmark, A., 2022. Temperature impacts on fish physiology and resource abundance lead to faster growth but smaller fish sizes and yields under warming. Global Chang. Biol., 28(21): 6239-6253.
Mariu, A., Chatha, A.M.M., Naz, S., Khan, M.F., Safdar, W., Ashraf, I., 2023. Effect of temperature, pH, salinity and dissolved oxygen on fishes. J. Zool. Syst., 1(2): 1-12.
Mba, O.I., Dumont, M.-J., Ngadi, M., 2015. Palm oil: Processing, characterization and utilization in the food industry–A review. Food Biosci., 10: 26-41.
Mitchell, S., Jr, J., 2011. Ammonia-Caused Gill Damage in Channel Catfish (Ictalurus punctatus): Confounding Effects of Residual Chlorine. Can. J. Fish. Aquat. Sci., 40: 242-247.
Mohammed, E.A.H., Ahmed, A.E.M., Kovács, B., Pál, K., 2025. The Significance of Probiotics in Aquaculture: A Review of Research Trend and Latest Scientific Findings. Antibiotics., 14(3): 242.
Navarro, R.D., de França, R.P., Paludo, G.R., Bizarro, Y.W.S., da Silva, R.F., Navarro, F.K.S.P., 2016. Physiological and hematological responses of Nile tilapia (Oreochromis niloticus) to different anesthetics during simulated transport conditions. Acta Sci. Technol., 38(3): 301-306.
Odo, J.I., Awua, Y., Gbargbar, M., Akange, E., 2023. Isolation and Identification of Bacteria from Ready-to-Eat Fish Sold in Makurdi Metropolis. PSM Microbiol., 8(3): 59-67.
Oluwatobi, A.A., Mutalib, H.A., Adeniyi, T.K., Olabode, J.O., Adeyemi, A., 2017. Possible aquaculture development in Nigeria: evidence for commercial prospects.
Omeji, S., Apochi, J., Egwumah, K., 2017. Stress concept in transportation of live fishes–a review. J. Res. For., Wildl. Environ., 9(2): 57-64.
Prein, M., Ahmed, M., 2000. Integration of aquaculture into smallholder farming systems for improved food security and household nutrition. Food Nutr. Bull., 21(4): 466-471.
Raj, A.J.A., 2008. Probiotic Performance on Fish Fry during Packing, Transportation Stress and Post-transportation Condition A. Jesu Arockia Raj,“A. Victor Suresh,” K. Marimuthu and” S. Appelbaum” The Bengis Centre for Desert Aquaculture. J. Fish. Aquat. Sci., 3(2): 152-157.
Ramlucken, U., Ramchuran, S.O., Moonsamy, G., Jansen van Rensburg, C., Thantsha, M.S., Lalloo, R., 2021. Production and stability of a multi-strain Bacillus based probiotic product for commercial use in poultry. Biotechnol. Rep., 29: e00575.
Ren, Y., Men, X., Yu, Y., Li, B., Zhou, Y., Zhao, C., 2022. Effects of transportation stress on antioxidation, immunity capacity and hypoxia tolerance of rainbow trout (Oncorhynchus mykiss). Aquac. Rep., 22: 100940.
Rollo, A., Sulpizio, R., Nardi, M., Silvi, S., Orpianesi, C., Caggiano, M., Cresci, A., Carnevali, O., 2006. Live microbial feed supplement in aquaculture for improvement of stress tolerance. Fish Physiol. Biochem., 32(2): 167-177.
Saba, A.O., Eyo, V.O., Elegbede, I.O., Fakoya, K.A., Ojewole, A.E., Dawodu, F.O., Adewale, R.A., Amal, M.N.A., 2024. Sustaining the blue bounty: Fish food and nutrition security in Nigeria’s evolving blue economy. AIMS Agric. Food., 9(2): 500-530.
Sampaio, F., Freire, C., 2016. An overview of stress physiology of fish transport: Changes in water quality as a function of transport duration. Fish Fish., 17.
Sanou, A., Coulibaly, S., Guéi, A.M.L., Baro, M., Tanon, E.F.T., Méité, N., Atsé, B.C., 2022. Assessment of some physico-chemical parameters of the fish farm water in Abengourou, Côte d’Ivoire. Egypt. J. Aquat. Biol. Fish., 26(5).
Shamsan, E.F., Ahmed, S., Khatoon, N., Ghoran, S.H., 2025. Nutritional Value of a Commercial Flathead Fish Species (Platycephalus indicus) from Karachi Waters, Pakistan. PSM Biol. Res., 10(1): 1-9.
Shamsan, E.F., Al-Jobory, H.J., 2018. Microbial status of sun-dried fish (Wazef) sold in different Yemeni Markets. PSM Biol. Res., 3(1): 1-8.
Shrivastava, J., Sinha, A.K., Cannaerts, S., Blust, R., De Boeck, G., 2017. Temporal assessment of metabolic rate, ammonia dynamics and ion-status in common carp during fasting: A promising approach for optimizing fasting episode prior to fish transportation. Aquac., 481: 218-228.
Tabassum, T., Sofi Uddin Mahamud, A.G.M., Acharjee, T.K., Hassan, R., Akter Snigdha, T., Islam, T., Alam, R., Khoiam, M.U., Akter, F., Azad, M.R., Al Mahamud, M.A., Ahmed, G.U., Rahman, T., 2021. Probiotic supplementations improve growth, water quality, hematology, gut microbiota and intestinal morphology of Nile tilapia. Aquac. Rep., 21: 100972.
Verma, S.A., 2006. Handbook of fisheries and aquaculture. ICAR.
Volkoff, H., Rønnestad, I., 2020. Effects of temperature on feeding and digestive processes in fish. Temperature (Austin, Tex.), 7(4): 307-320.
Wang, J., Xu, K., Chen, X., Wang, H., Li, Z., 2024. Effects of Transport Stress (Duration and Density) on the Physiological Conditions of Marbled Rockfish (Sebastiscus marmoratus, Cuvier 1829) Juveniles and Water Quality. Fishes., 9(12): 474.
Yaka, D.A.M., Tiemeni, A.A., Zing, B.Z., Nenkam, T.L.L.J., Aboubakar, A., Nzeket, A.B., Tcholong, B.H.F., Mewouo, Y.C.M., 2020. Qualité physico-chimique et bactériologique des eaux souterraines et risques sanitaires dans quelques quartiers de Yaoundé VII, Cameroun. Int. J. Biol. Chem. Sci., 14(5): 1902-1920.
Yaqub, S.S., Hadyait, M.A., Salim, S., Bhatti, E.M., Ullah, M.Z., Begum, I., 2017. Risk Assessment of Salmonella spp. in Labeo rohita Marketed in Lahore, Pakistan. PSM Microbiol., 2(3): 74-77.